A new JBJS report compares arthroscopic versus open ankle arthrodesis. The authors analyzed long-term patient-reported outcomes, major complications, and reoperations. JBJS Editor-in-Chief Dr. Marc Swiontkowski
Tag: fusion
Co-author Alfred J. Pisano, MD selects the 5 most intriguing findings from the recently published “What’s New in Spine Surgery.” Cervical Spondylotic Myelopathy – A recent
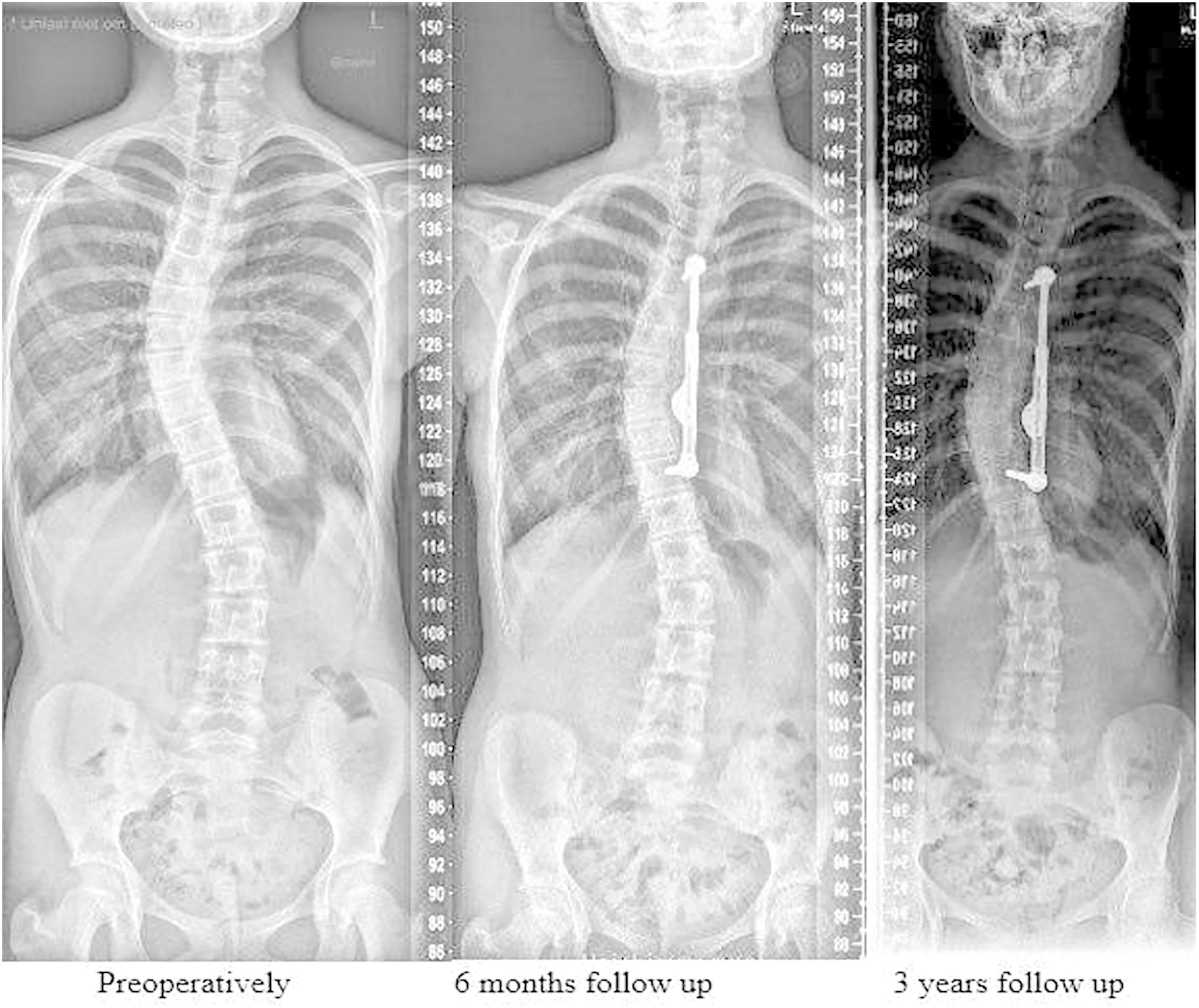
As we continue to evaluate methods of treating adolescent idiopathic scoliosis (AIS), growth modulation has become a hot topic. Such techniques aim at curve correction without the need for fusion, and can involve either tethering on the convex side of the curve or distraction on the concave
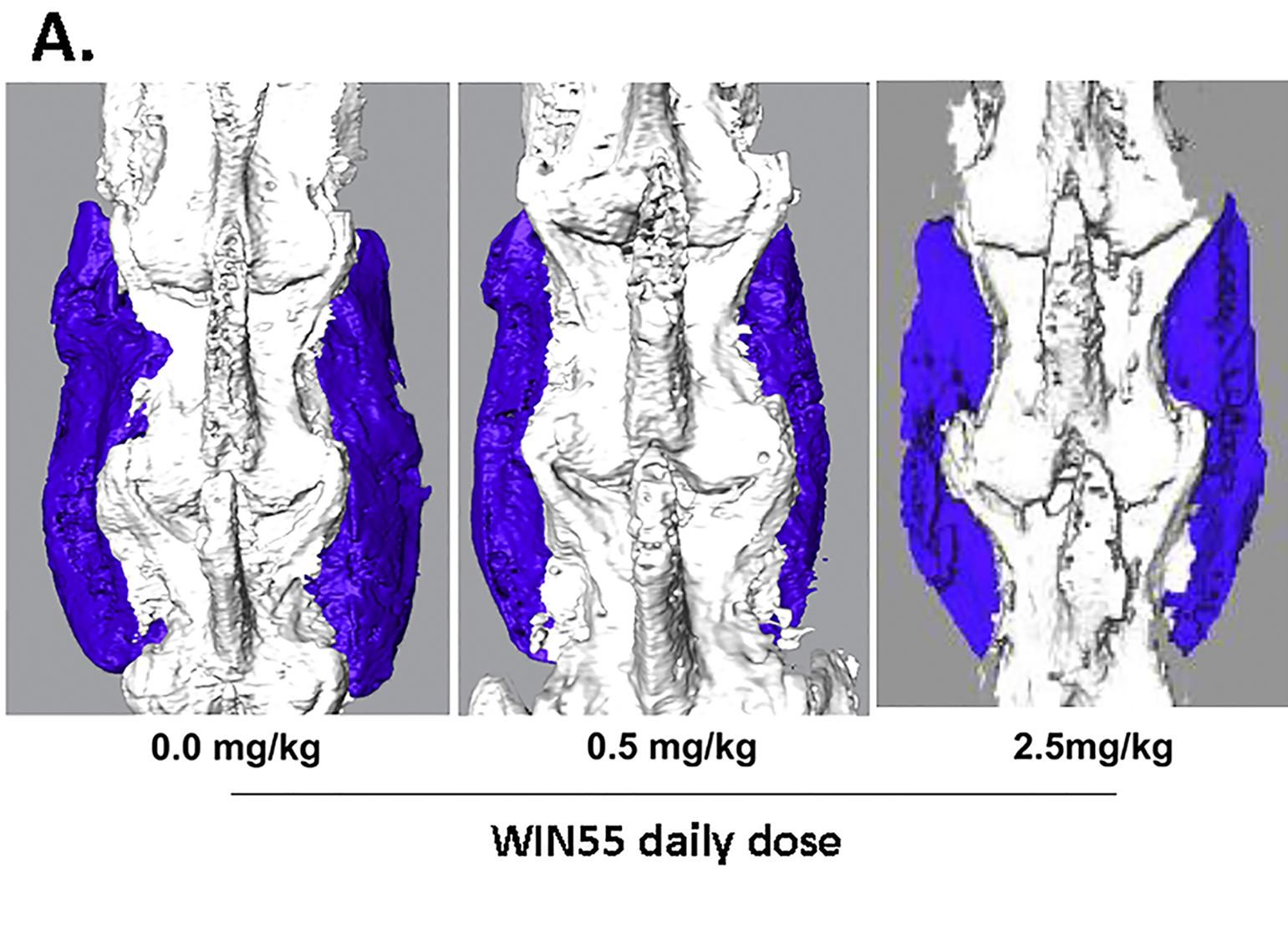
The dangers of chronic opioid use have rightly been at the forefront of orthopaedic practice considerations in recent years. The widespread use of regional anesthesia and periarticular-injection cocktails, targeted NSAID utilization, and strict limitations on opioid use
In the October 7, 2020 issue of The Journal, Du et al. report on a multicenter database-derived cohort of 167 patients with early-onset scoliosis treated
Donor-site morbidity from harvesting autologous bone graft has driven the decades-long search for a substitute that performs at least as well as a patient’s own
Health-related quality of life (HRQOL) in adulthood is an important outcome measure for patients diagnosed with juvenile or adolescent idiopathic scoliosis. In the May 16,
In the December 7, 2016 issue of JBJS, Krause et al. analyze data from a 2013 industry-sponsored RCT to investigate correlations between nonunions of hindfoot/ankle
A therapeutic Level II study by DiGiovanni et al. in the August 3, 2016 edition of The Journal of Bone & Joint Surgery examined the
In the December 16, 2015 edition of The Journal, Pellegrini et al. present the results from a cohort of 23 patients who had initially undergone

