New findings in spine surgery are covered in the latest JBJS Guest Editorial What’s New in Spine Surgery. Here, we summarize the 5 most insightful
Category: Spine
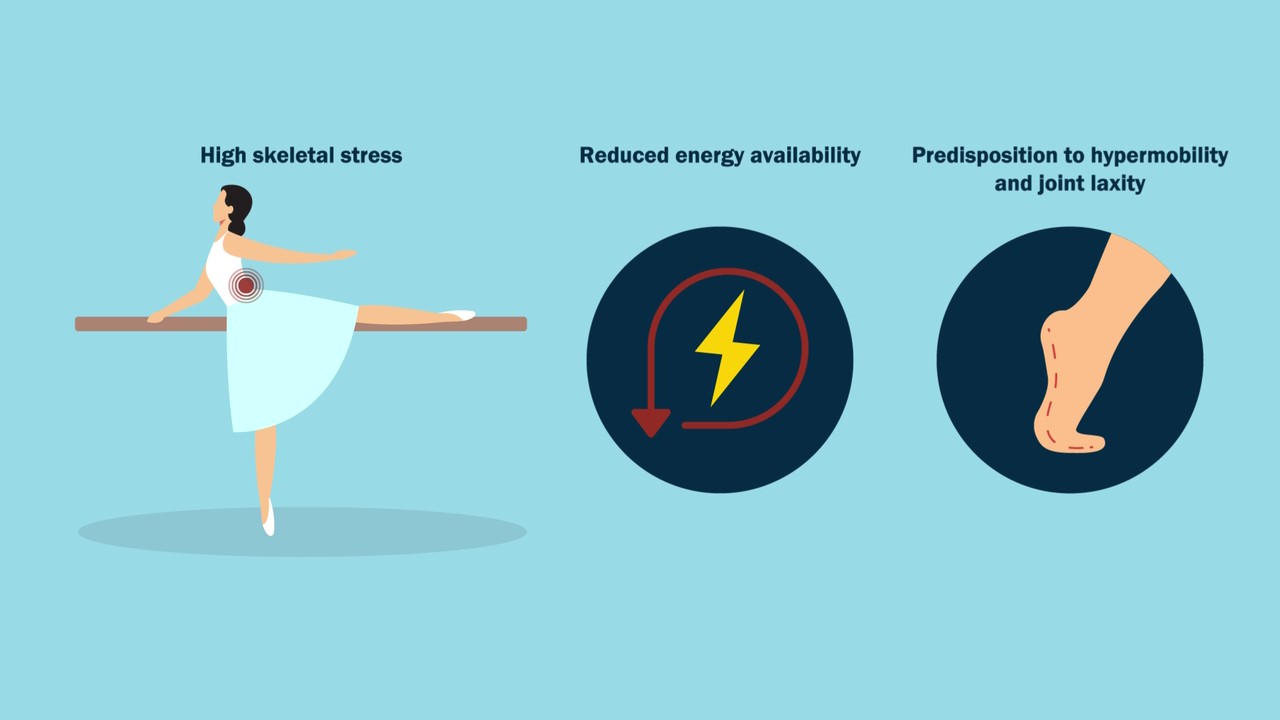
Dr. Matt Schmitz, JBJS Deputy Editor for Social Media, offers this post on 2 new studies now available at JBJS.org. As a pediatric orthopaedic surgeon
Notable findings in pediatric orthopaedics are presented in the JBJS Guest Editorial What’s New in Pediatric Orthopaedics. Here, we summarize the 5 most impactful studies,
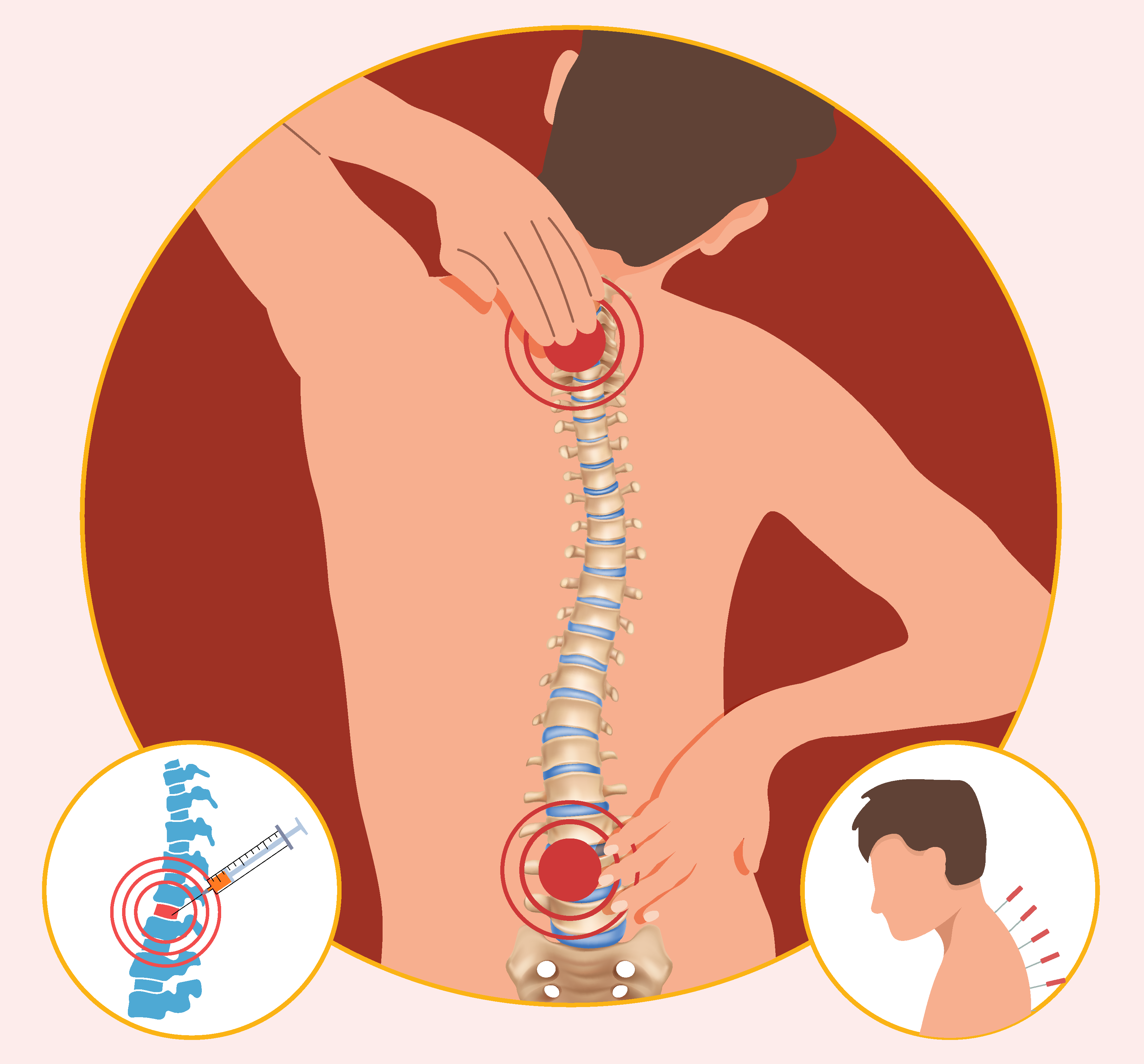
This post spotlights 2 new studies in JBJS that examined outcomes of spine surgery using nationwide databases. Patients with degenerative lumbar disease may turn to
Dr. Matt Schmitz, JBJS Deputy Editor for Social Media, discusses 3 recent studies that reflect the rigorous standards, and challenges, of randomized clinical trials (RCTs).
Investigators in France evaluated the long-term outcomes of 1- and 2-level total disc arthroplasty (TDA) in patients with chronic lumbar degenerative disc disease. A total
For patients with adolescent idiopathic scoliosis (AIS), could certain aspects of the spine examination be reliably conducted via telemedicine? Dr. Matt Schmitz, JBJS Deputy Editor
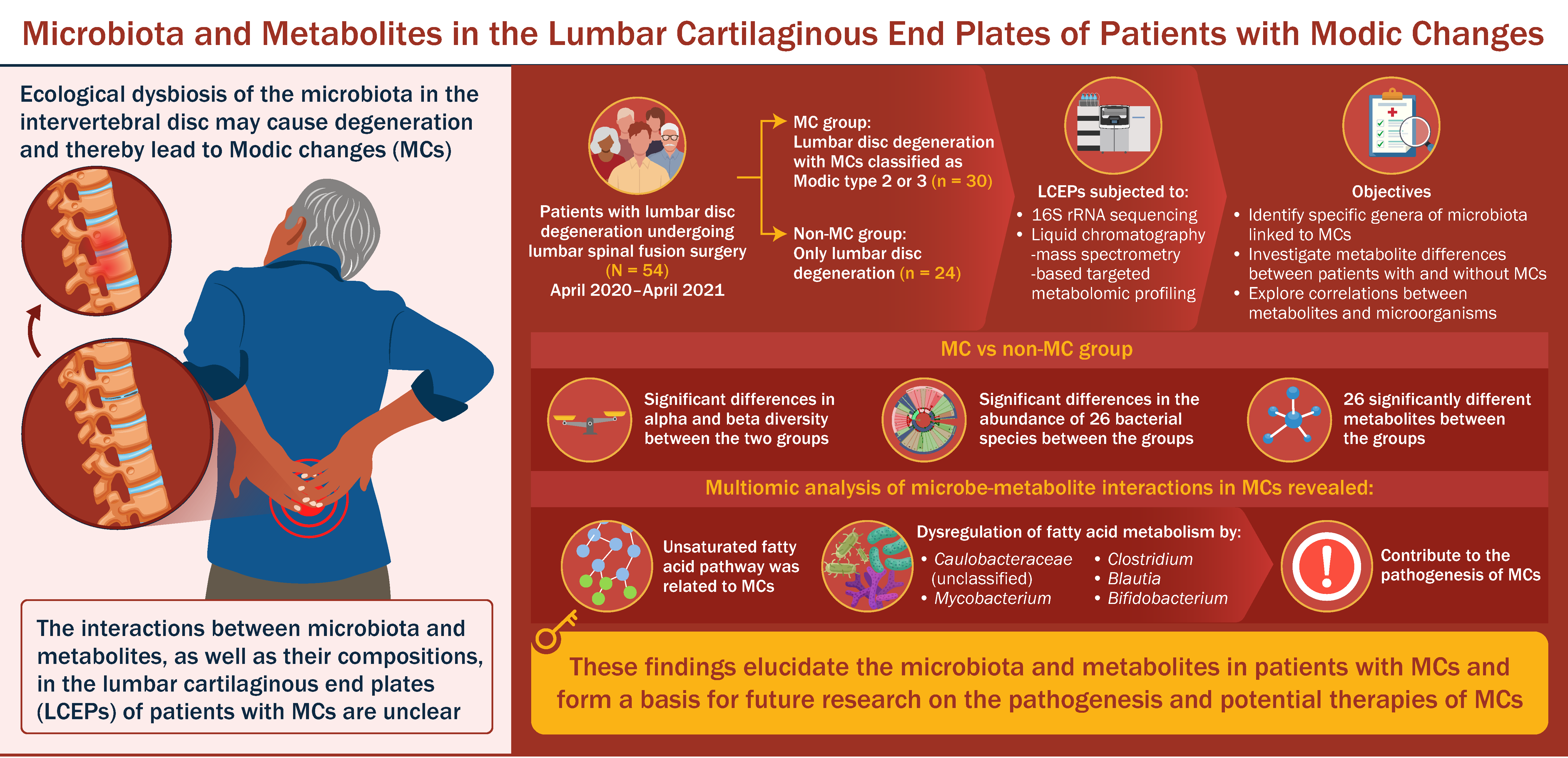
As reported recently on OrthoBuzz, a study by Nian et al. provides novel exploration of microbiota and metabolites in patients with Modic changes—changes in vertebral
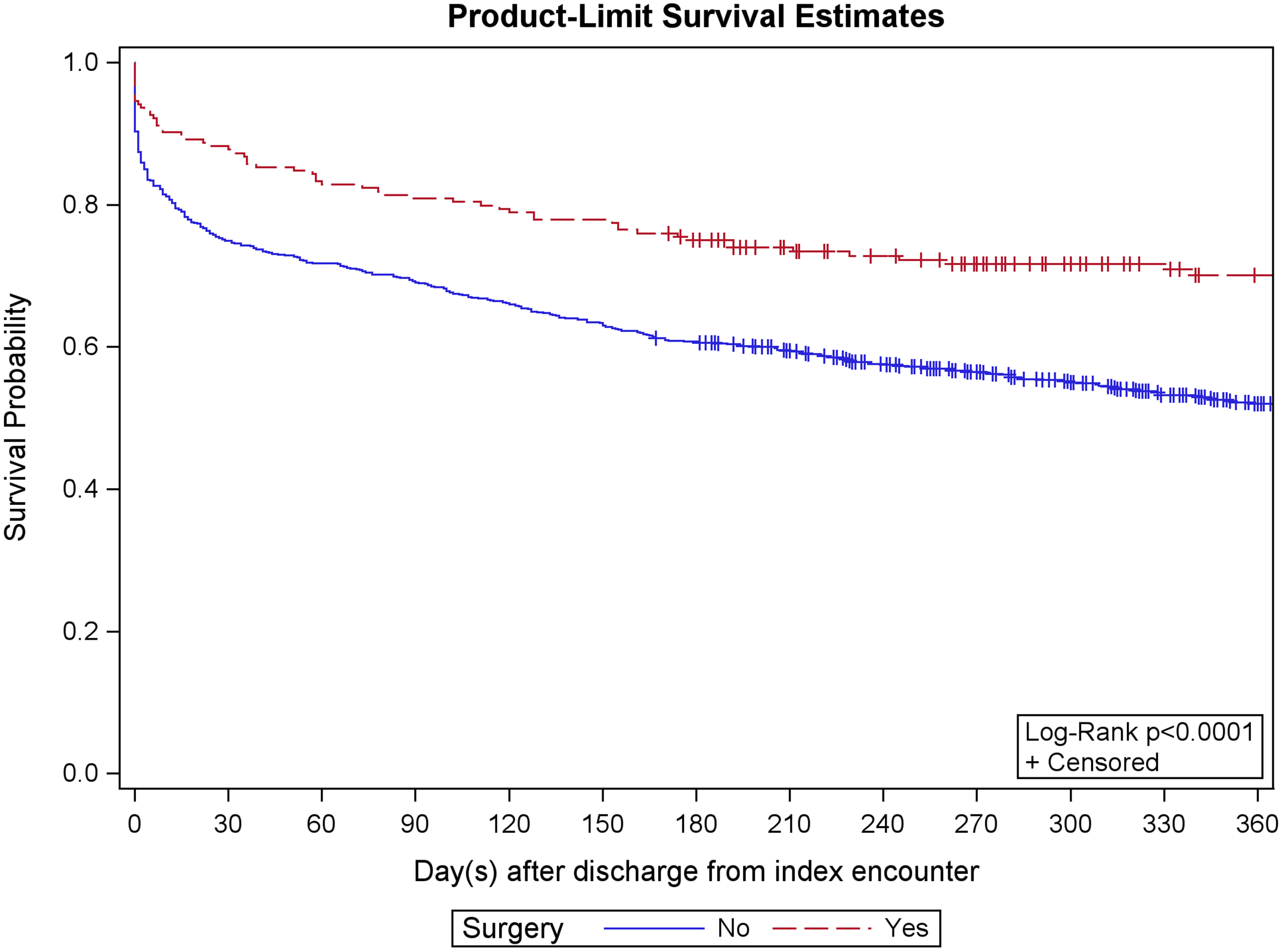
In a new study in JBJS, investigators found that surgical intervention “may confer a survival benefit” in elderly patients with dementia who sustain a type-II
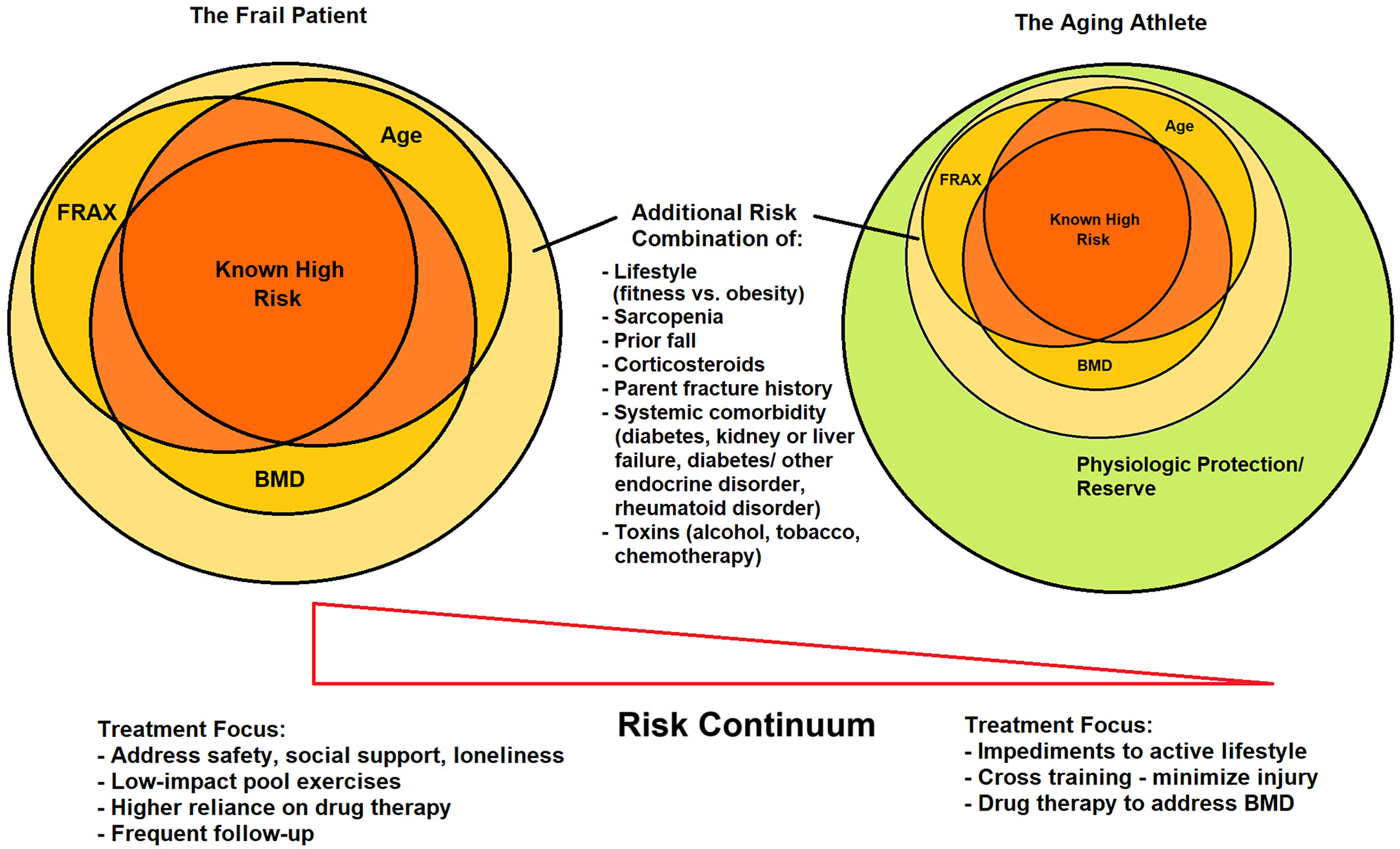
New insights regarding osteoporosis treatment and fracture prevention are highlighted in the recent JBJS Guest Editorial What’s New in Osteoporosis: Emphasis on the Aging Athlete.