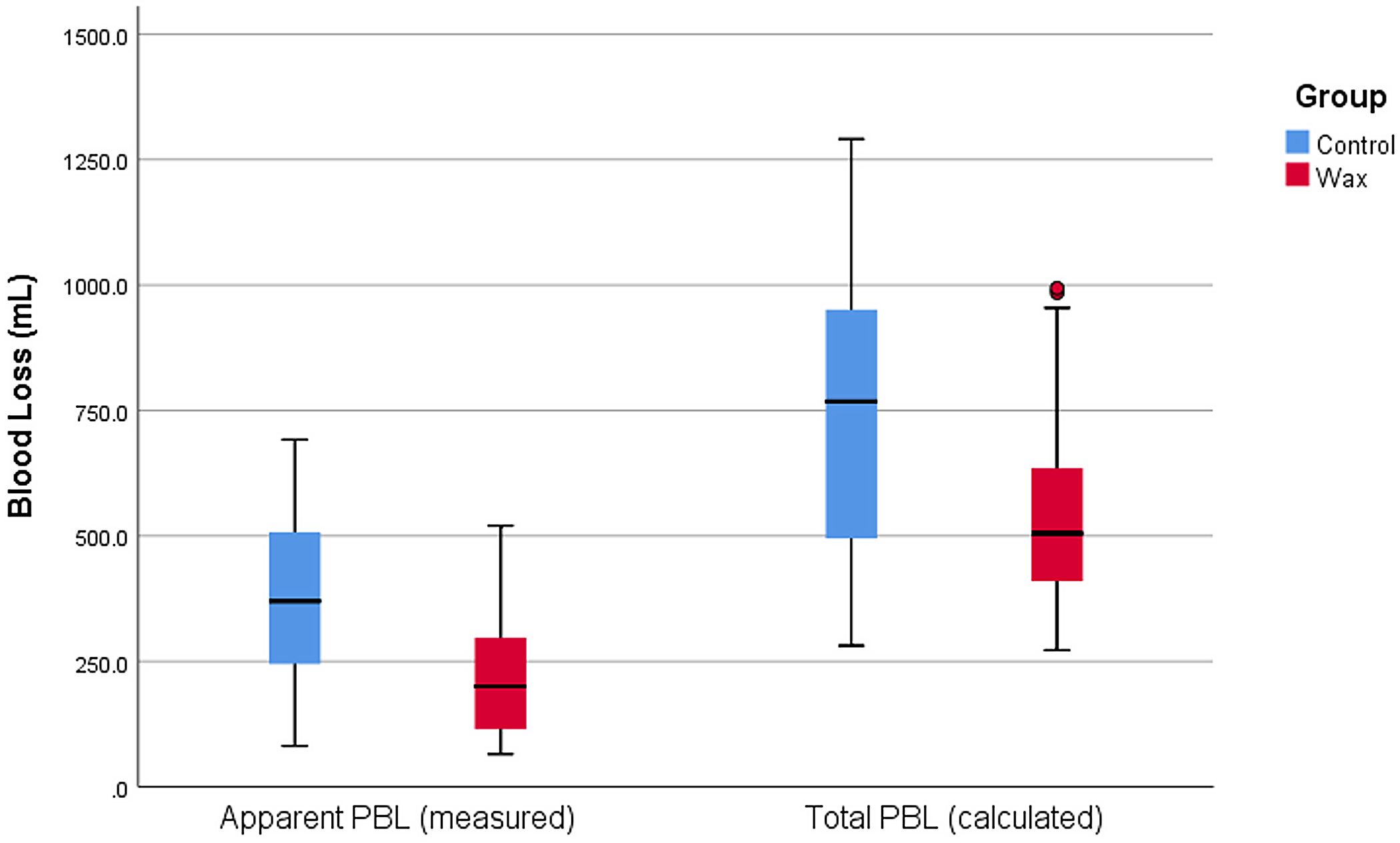
A number of measures have been suggested for reducing perioperative blood loss (PBL) in total hip arthroplasty (THA). A new randomized clinical trial in JBJS
Tag: total hip arthroplasty
In the current issue of JBJS, Goh et al. report on the association between glucose variability and postoperative complications following aseptic revision total joint arthroplasty
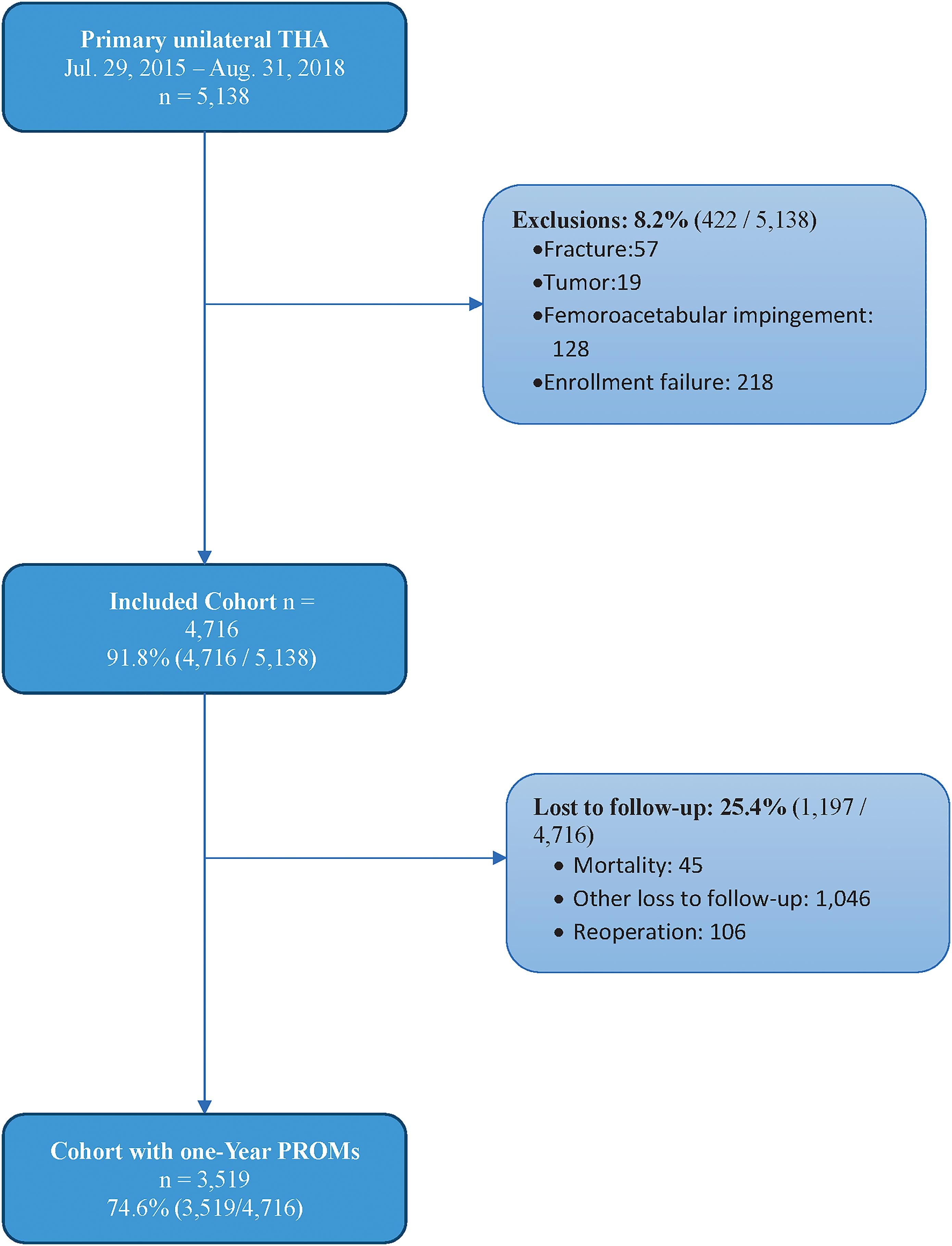
Total hip arthroplasty (THA) is one of the most successful procedures in not only orthopaedics but all of surgery. With the dramatic rise in the number of THAs performed annually and the necessary focus on value-based
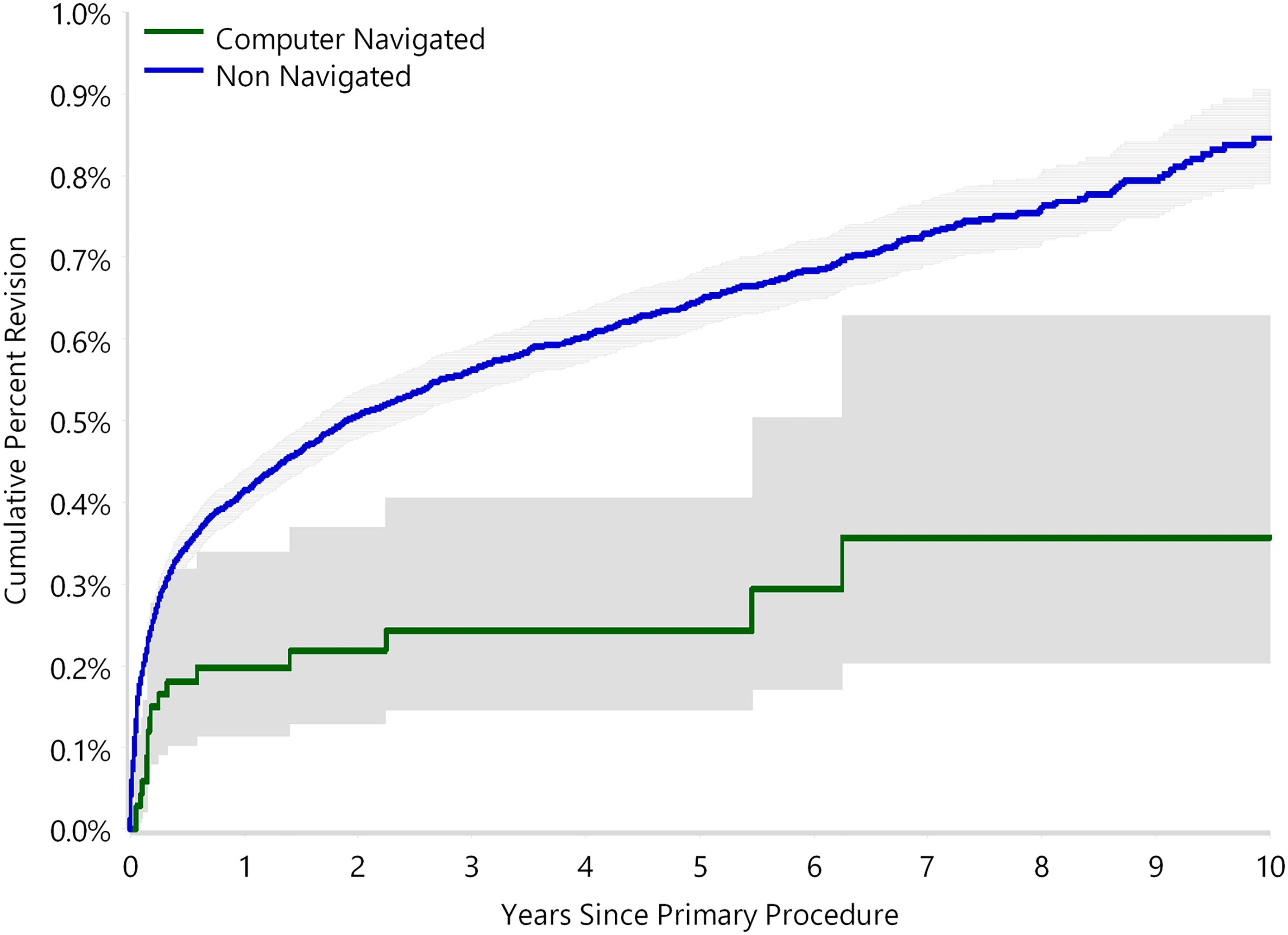
The use of computer navigation in total joint arthroplasty is increasing. Navigation has been employed less in total hip arthroplasty (THA) than in total knee arthroplasty, perhaps because of the increased need for
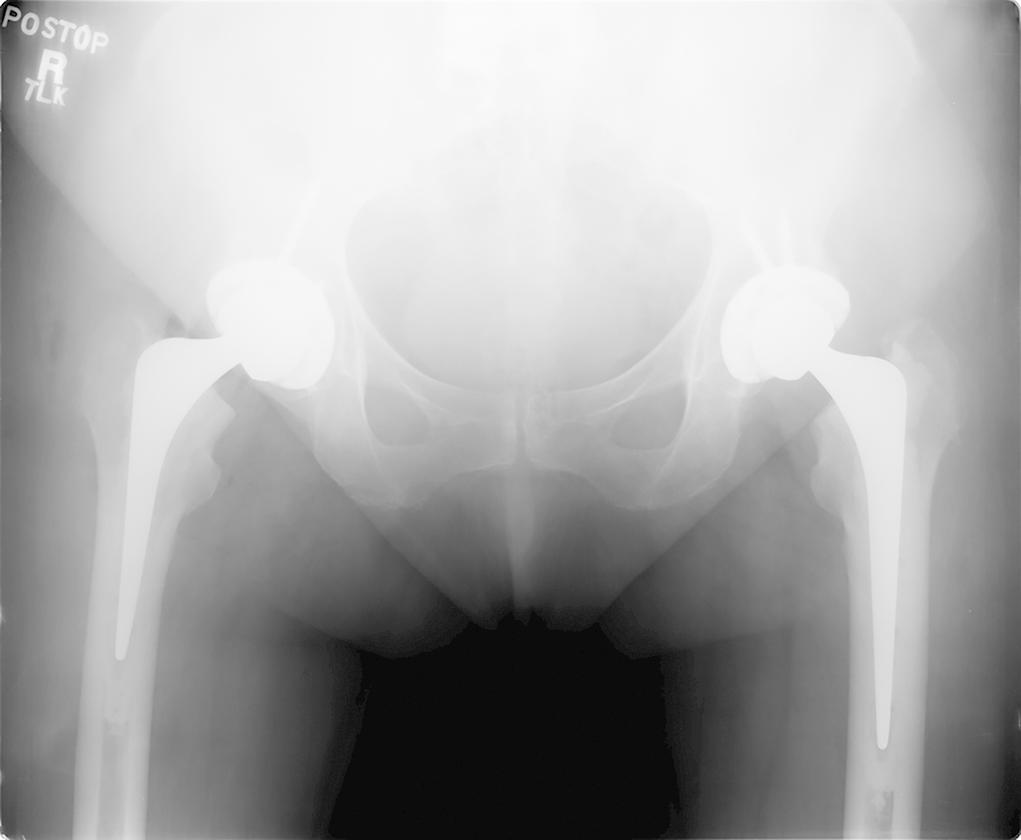
In a new study reported in JBJS, Gausden et al. evaluated implant survivorship, complications, and radiographic results of a specific collarless, polished, tapered cemented stem (Exeter; Stryker) used selectively in a predominantly elderly
Every month, JBJS publishes a review of the most pertinent and impactful studies from the orthopaedic literature during the previous year in 14 subspecialty areas. Click here for
Genetic susceptibility to orthopaedic conditions is of interest to clinicians and patients alike. While the link between genetics and certain pediatric conditions is known, studies
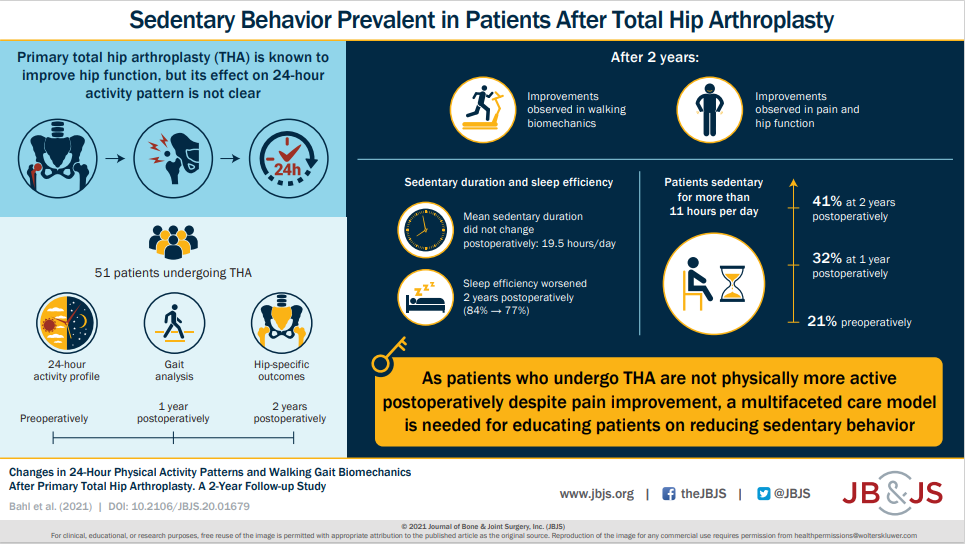
Together with improvements in self-reported pain and perceived physical function, patients had significantly improved gait function postoperatively. Read the full article here.
In a prospective case-control study reported in JBJS, Ohmori et al. evaluated factors related to postoperative gait speed in patients with osteoarthritis undergoing total hip arthroplasty. They found that the preoperative, contralateral-side OLST (one-leg standing time) was a significant factor
As the volume of total hip arthroplasty (THA) cases continues to rise, so too will the need for revision surgery. Revision THA can be complicated by insufficient bone stock on either the femoral or acetabular side, and researchers are gaining further insight into bone