This week on OrthoBuzz, JBJS is pleased to present another Elite Reviewer profile. The Elite Reviewers Program recognizes our best reviewers for their outstanding efforts.

In a recent article in JBJS, Escalera et al. provide an informative overview of the American Association of Latino Orthopaedic Surgeons (AALOS). Since its founding

Key findings in sports medicine on topics such as rotator cuff repair, anterior cruciate ligament (ACL) reconstruction, and more are presented in the new JBJS

OrthoBuzz marks the 1-year anniversary of JBJS OrthoCorps, an initiative that aims to preserve the voices and memories of the global orthopaedic community. In March

This week on OrthoBuzz, JBJS is pleased to present another Elite Reviewer profile. The Elite Reviewers Program recognizes our best reviewers for their outstanding efforts.

Interested in being published on the JBJS OrthoBuzz blog? Residents and trainees are invited to submit a post to Resident Roundup. Share your experience as
Dr. Matt Schmitz, JBJS Deputy Editor for Social Media, discusses a new study on the impact of robotic assistance and computer navigation on periprosthetic joint

This week on OrthoBuzz, JBJS is pleased to present another Elite Reviewer profile. The Elite Reviewers Program recognizes our best reviewers for their outstanding efforts.
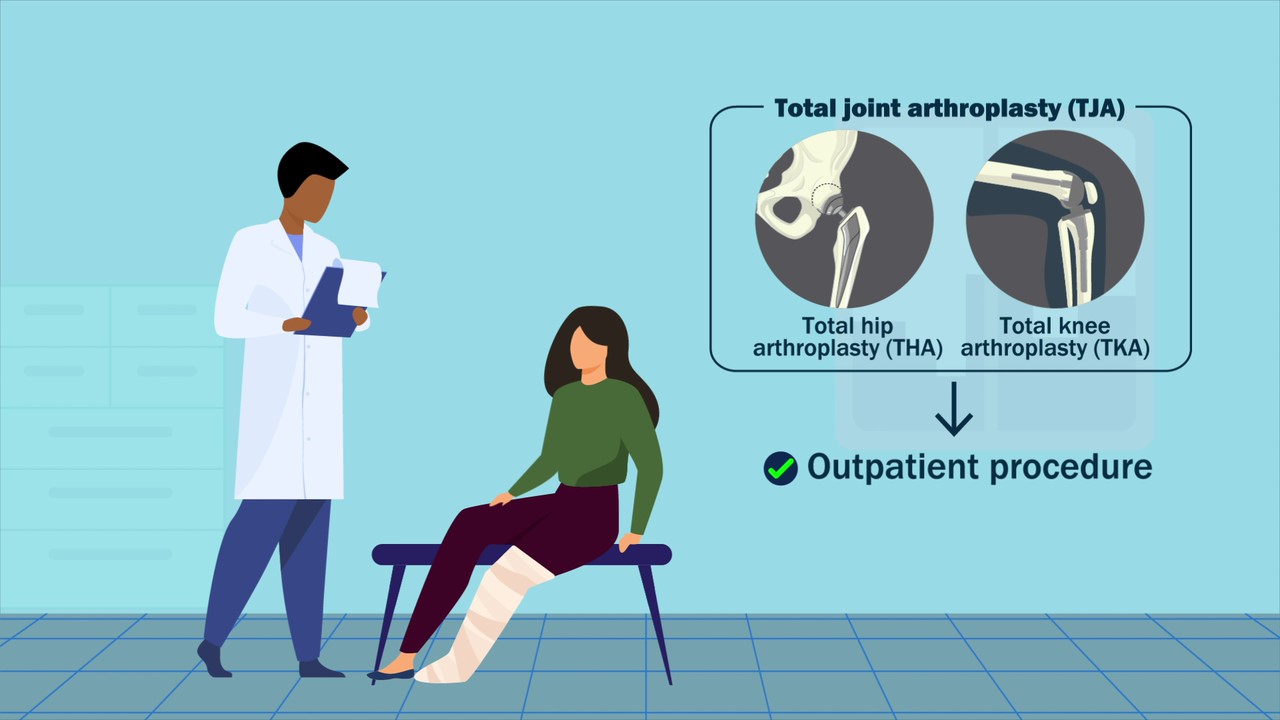
JBJS Editor-in-Chief Dr. Marc Swiontkowski shares his thoughts on a new study showing the potential of preoperative bladder scanning to predict postoperative urinary retention in

This week on OrthoBuzz, JBJS is pleased to present another Elite Reviewer profile. The Elite Reviewers Program recognizes our best reviewers for their outstanding efforts.

