Vitamin D supplementation, fracture risk, and osteoporosis drug therapies are among several topics explored in recent studies featured in the new JBJS Guest Editorial: What’s
Search Results for: fragility fracture
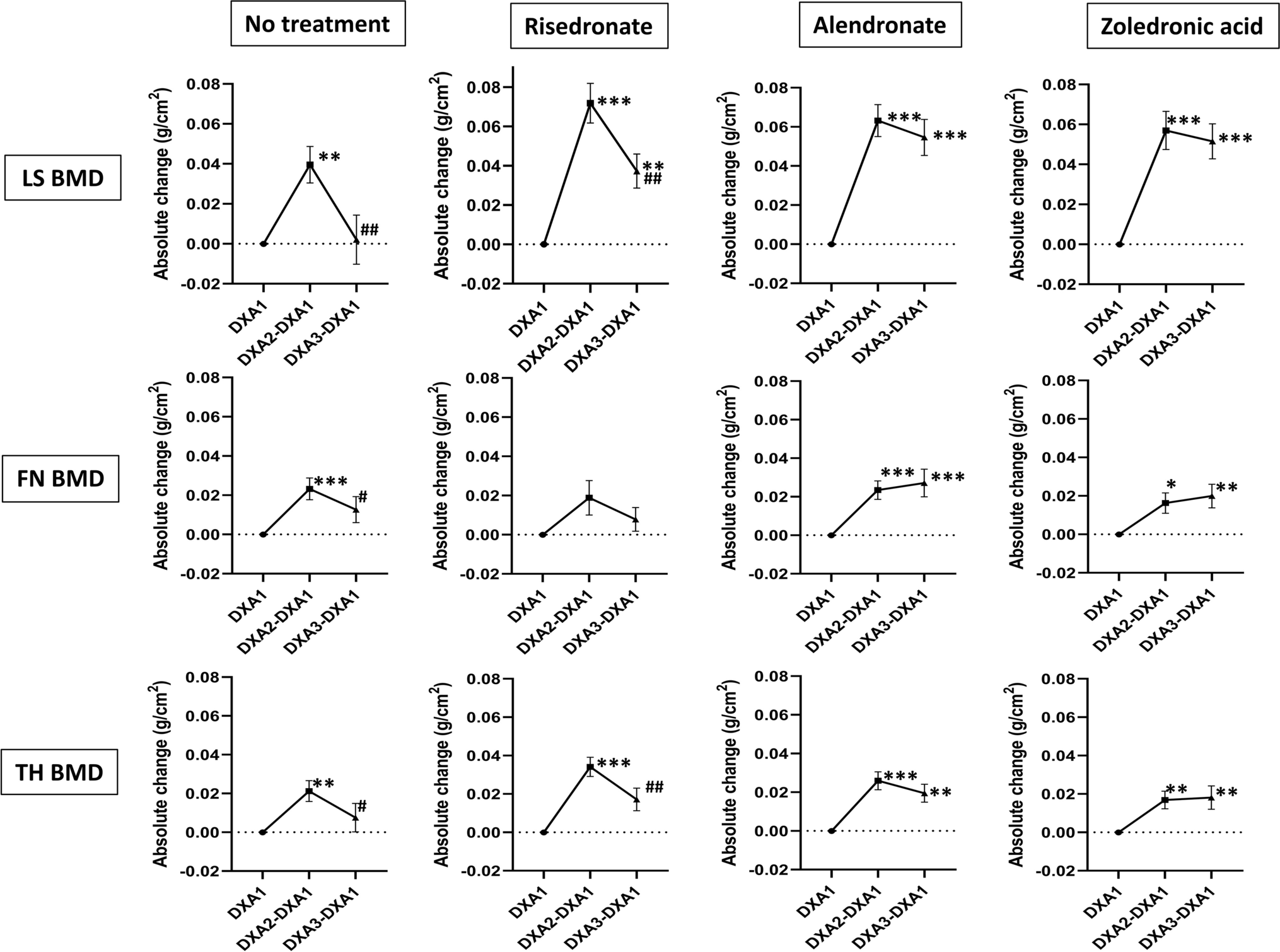
The recent JBJS Guest Editorial “What’s New in Osteoporosis and Fragility Fractures” provides an update on this important area of orthopaedic research. The authors review
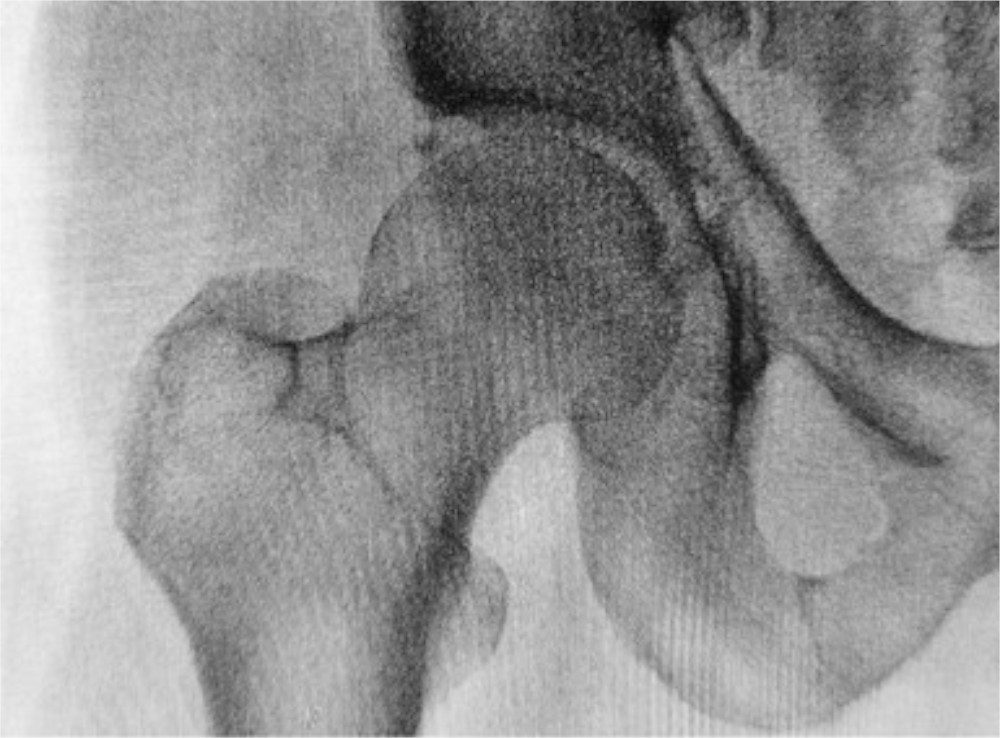
JBJS Editor-in-Chief Dr. Marc Swiontkowski reflects on a new study examining changes in skeletal muscle mass following femoral fragility fracture. Since the early 20th century,
After nearly 2 decades, the orthopaedic community has made a good start on assuming our responsibility in the diagnosis of osteoporosis after a patient’s initial low-energy fracture. We are seeing a positive impact from programs such as the American Orthopaedic Association’s “Own the Bone” initiative as well
OrthoBuzz occasionally receives posts from guest bloggers. This guest post comes from James Blair, MD, in response to a recent edition of the OrthoJOE podcast. Geriatric
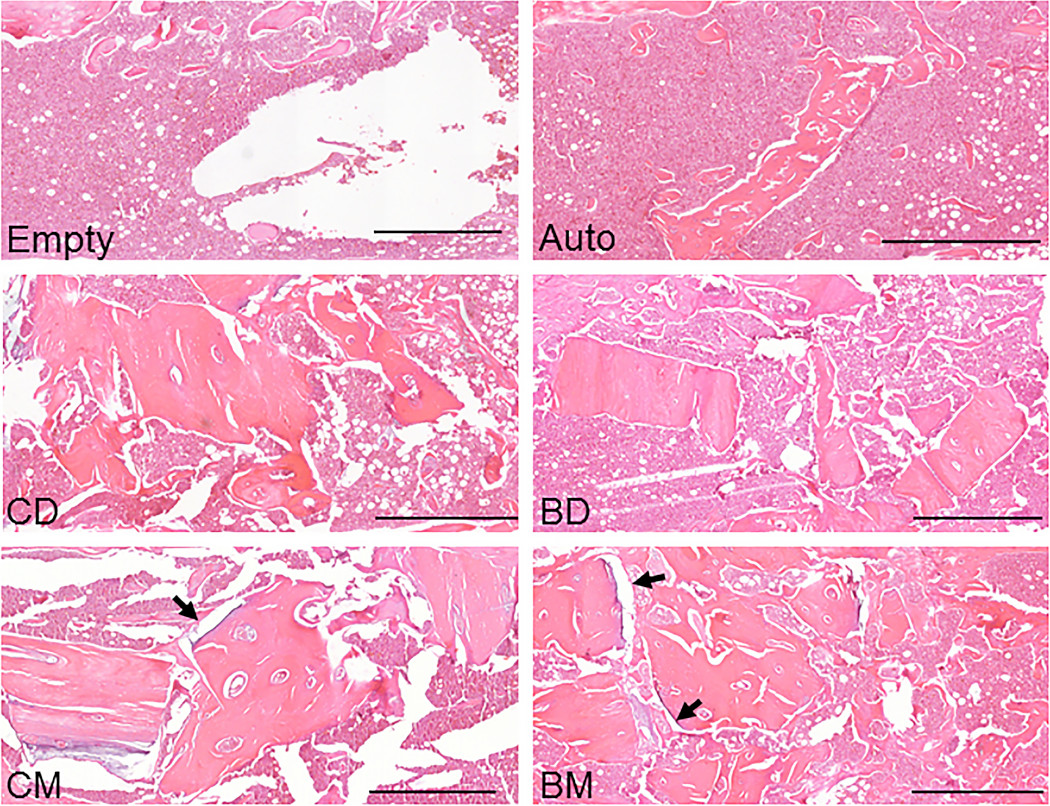
The findings of a new animal study suggest that bisphosphonate treatment in donors may indeed be relevant when mineralized allografts are used in orthopaedic procedures.
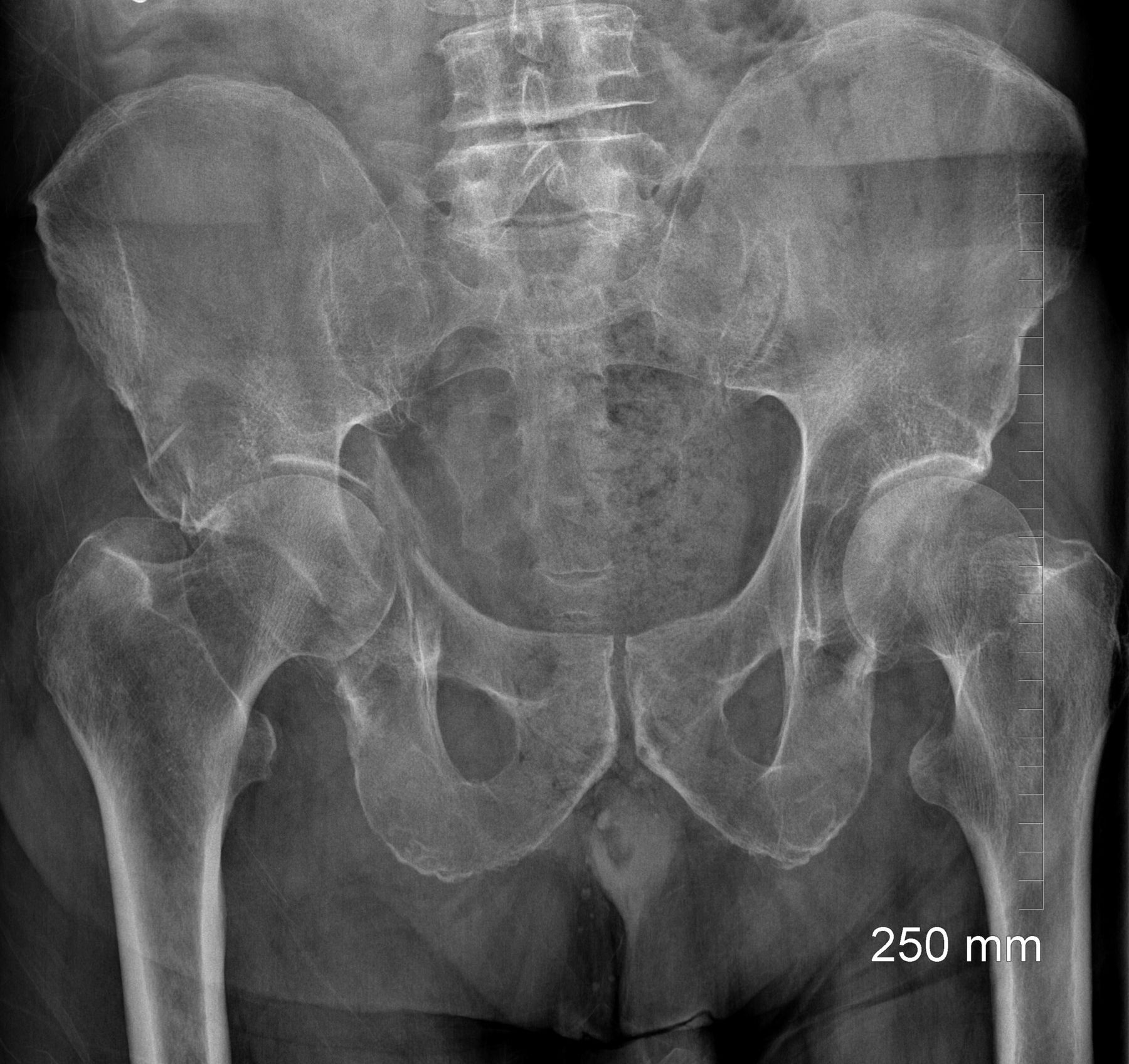
When discussing patient outcomes of hip fractures, we often are speaking of fractures of the proximal part of the femur. But what about the other side of the hip joint, the acetabulum? Among fragility-related injuries, we’re seeing a rise in the incidence of acetabular fractures. And

Members of the orthopaedic community worldwide turn to JBJS to stay informed of current evidence, techniques, and the latest research in musculoskeletal care. Of the
Osteoporosis is the major contributor to the increasing incidence of fragility fractures associated with low-energy falls. The other contributor is the populous baby-boomer generation that
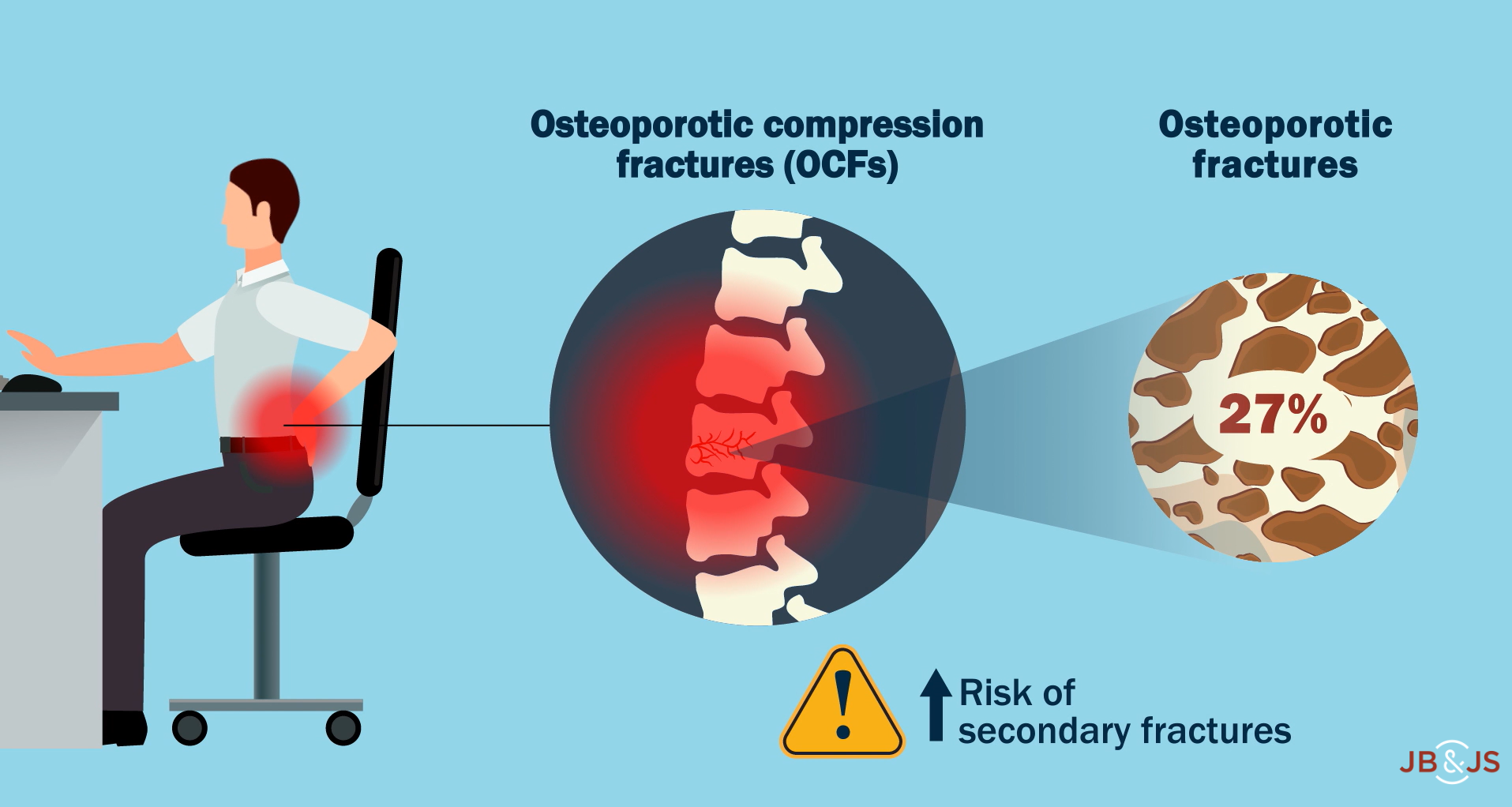
In a new study in JBJS, Mills et al. evaluate whether cement augmentation increases the risk of secondary fracture following a vertebral osteoporotic compression fracture